Design dynamic Field Medical teams for optimal medicine adoption
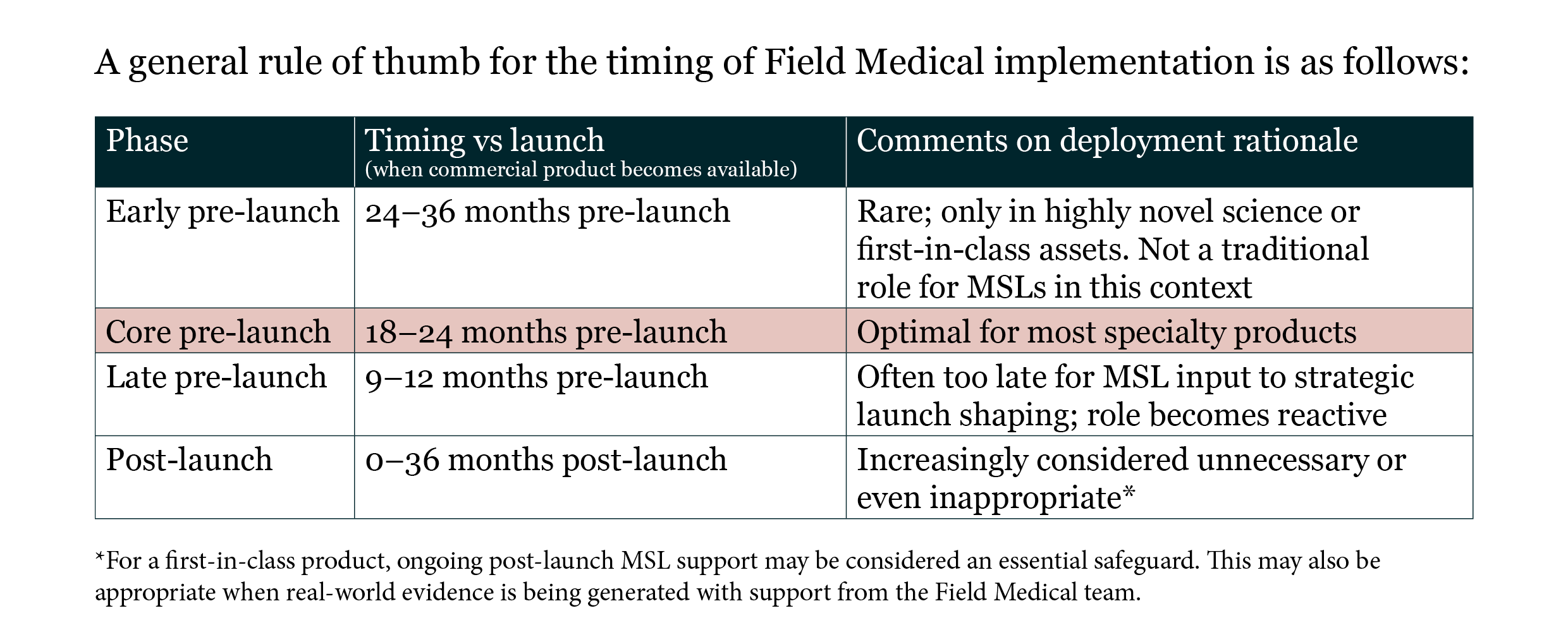
The implementation of a field-based medical service (usually a medical science liaison [MSL] team) is widely regarded as a must-have in the early stages of product launch readiness – whether to support the delivery of Phase 3 clinical trials, initiate early external engagement with potential future prescribers and key opinion leaders (KOLs), or begin Medical Affairs initiatives.
When executed well, it can shape external perception, drive evidence adoption and build long-term credibility. Done poorly, it risks becoming an ineffectual, expensive, quasi-commercial function that delivers limited strategic value and may introduce compliance risk.
At a basic level, any Field Medical team can be recruited, trained and tasked with the following generic list of responsibilities:
Clinical trial support at Phase 2, 3 and 4
Site identification
Feasibility feedback
Investigator relationships
Data interpretation dialogue (post readouts)
2. KOL mapping and segmentation
National, regional and emerging voices
Academic vs community
Guideline influencers
Payer-facing opinion leaders (if relevant)
3. Insight generation and advisory boards
Current treatment paradigms, infrastructure and service readiness
Barriers to adoption
Clinical scepticism
Evidence gaps
Perceived positioning vs competitors
4. Scientific exchange and scientific communication
Disease state education and unmet need
Education on mechanism of action of the product
Reactive pipeline communication
Congress engagement follow-up
5. Evidence planning support
Identify unmet data needs
Inform real-world evidence planning
Shape post-marketing study concepts
Engage with KOLs to support them with investigator-initiated studies
However, how this generic list of activities is implemented, the timing of execution and the level of specificity in plans for the Field Medical function vary widely across companies, regions and products.
In this article, we outline a structured and rational approach to designing a new Field Medical team and introduce the concept of a Dynamic Field Medical Model, or DFMM, as a framework to maximise impact for both newly established and existing field-based medical teams.
Rational approach to design a new Field Medical team
The aforementioned list of Field Medical responsibilities is commonly used as a template for hiring and deploying a Field Medical team. However, it remains just that – a template. As such, it offers an inadequate level of customisation for what represents a significant organisational investment, both in time and costs.
A better way of approaching the question of how best to hire and implement a Field Medical team is to start with a detailed environmental analysis, that defines:
Disease prevalence
Treatment centre distribution, capability and capacity
Number of priority KOLs and healthcare professionals (HCPs)
Geography size and country-specific challenges
Complexity of the disease process and patient pathways, as well as the product data.
Assuming the answers to these questions are clear, designing a Field Medical team tailored to this environment would be a reasonable thing to do. However, in most cases, particularly if the company is new to the geography, the disease area or both, the first step in Field Medical deployment would be to gain crystal clarity on this list.
This clarity can be achieved through a sequence of initiatives, either led internally or facilitated by a consultancy partner. Better still, it can be delivered by a single, strategically minded Field Medic with experience in team design and expertise in operational effectiveness. This individual becomes your first hire in the field and is tasked with executing a three-phase approach:
1. Complete an initial analysis of the environment per the categories above
2. Design a bespoke Field Medical team that exactly addresses the specific challenges presented, acknowledging that these may evolve with further insights
3. Hire and lead the team
Phase 1 should take no longer than 3–6 months, and once the answers to the environmental analysis are clear, then the Field Medical Lead can develop an evidence-based resource plan to hire and deploy Field Medics that exactly fit the challenges of the environment that have been identified. Once approved by senior leadership, Phase 3 involves hiring team members based on bespoke job descriptions (derived from the outcomes of Phase 1) and subsequently leading this team.
The Dynamic Field Medical Model
If the guidance above has been followed, then by definition, the Field Medical team will:
1. Be exactly matched in size (i.e. the right number of Field Medical associates), form (i.e. team structure and reporting lines) and function (i.e. skills, capabilities and priorities) to the challenges facing the product at the specific time and place of its launch.
2. Operate in a rapidly changing environment, shaped by emerging data (both product and disease area), changing timelines for regulatory approval, reimbursement and launch, rapid upscaling of internal resourcing (including the emergence of a Commercial Brand team and sales-team), and inevitable external factors such as competitor progress – all ensuring that no month looks quite like the one before.
Ironically, within this fluidity, many companies choose to hire a fixed number of Field Medical associates and assign a fixed set of activities. Some of these, such as engagement with trial sites, may remain consistent over the course of months and years, but others – specifically external engagement, insights gathering and data dissemination – will need to change repeatedly, rapidly and responsively to this changing environment.
The concept of a DFMM has been discussed informally for a number of years. In essence, it requires organisations to reassess their Field Medical team and activities on a regular basis (usually quarterly), evaluating structure and function against the evolving external environment and anticipated changes – both internal and external.
Crucially, this analysis starts from an assumption that the form and function of the Field Medical resource may no longer be optimal. The exercise seeks to disprove this hypothesis and, if unable to do so, the organisation must acknowledge that adjustments are needed – often in function, but sometimes in form too.
In its most basic form, this prevents a typical error in the deployment of a Field Medical team – namely, a non-promotional scientific field force that continues to undertake external engagement activities many months or years after launch. When used appropriately, however, this DFMM can also result in the regular tuning and optimisation of field activities, ensuring that no two quarters look quite the same.
This personalisation of a team’s function ensures high levels of impact against agreed key performance indicators (KPIs), while also encouraging team members to think proactively about their work each week. Field Medical team members who actively seek such a challenge value the ever-changing list of priorities and the critical thinking that is required to match priority activities to external environmental needs.
Compliance
No design of an initial external engagement resource would be complete without considering the inherent compliance and legal risks. Nowhere is the precipitous nature of conversion from R&D activities into commercialisation felt more strongly than in the 6-month period pre-product launch. During this time, Field Medical teams face high scrutiny – from regulators, competitors and sometimes HCPs. Regulatory engagement at launch can significantly impact execution if not carefully managed.
In the pre-approval phase, guardrails around proactive scientific exchange are critical, particularly given the differences between US and EU regulations.
Key considerations are:
Having clear separation from Commercial
Maintaining a medical reporting line
Handling specific and non-promotional materials only
Adhering to standard operating procedures (SOPs) governing proactive vs reactive discussions
Undertaking training on off-label and pre-approval communications
The impact that Field Medical teams can have in the pre-launch and launch phase of any new medicine is second to none. All companies should place the development of such an external engagement force at the very top of their launch readiness plans.
However, the level of personalisation within these Field Medical teams is typically low. Many companies simply hire an arbitrary number of MSLs and hope that their activity will deliver meaningful results.
By taking a thoughtful and rational approach to designing a new Field Medical team, and then routinely assessing it against the evolving external and internal needs of the product using the DFMM, the Field Medical team can be built for maximum effect and continually tuned for impact. The benefits that this approach delivers for the product, Commercial teams, HCPs and ultimately patients should not be underestimated.
Authored by:
Dr. Matthew Goodman MB.ChB, MFPM, DPM, FRSM
CEO, LUCENT
To implement the Dynamic Field Medical Model in your organisation, contact Matthew Goodman or Trevor Brett.